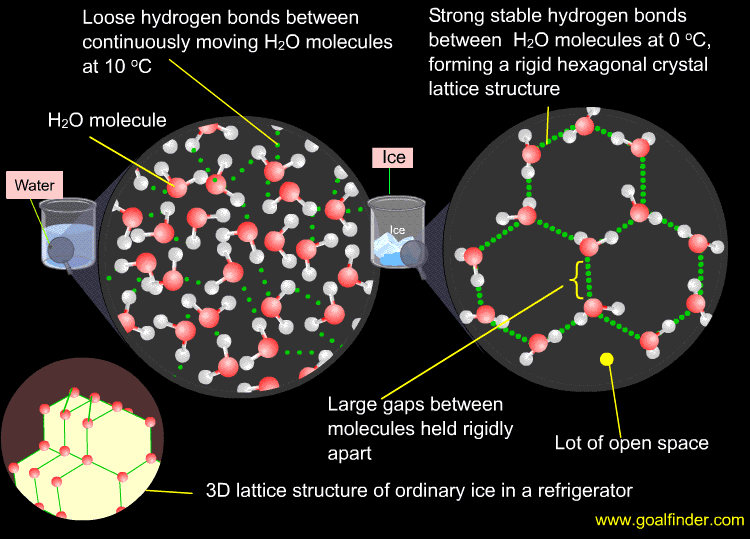
Density of Water
As liquid water cools, the molecules come closer together and become more dense. Water is most dense at 4 degrees Celsius, but the molecules are still moving (not frozen). At temperatures below 4 degrees Celsius, there is only virational movement as the water begins to freeze and take on a less dense lattice structure. This is why a can of soda will burst when placed in a freezer. The water expands as it freezes and breaks the can.
This important characteristic of water is vital to life on earth. Since ice is less dense than water, ice will float on top of water. If it did not float, it would sink and ponds, lakes, rivers and even the ocean would freeze solid thereby making life impossible in teh water. Since scientists believe that life first originated in the oceans, this would mean that early life could not have developed and moved to onto land. Additionally, it is the life in the water that sustains life on land, through the oxygen producing algae to the food sources such as fish.
When a body of water freezes on the surface, the ice acts as an insulator to prevent the water below it from freezing. This protects aquatic organisms so that they can survive the winter.
This important characteristic of water is vital to life on earth. Since ice is less dense than water, ice will float on top of water. If it did not float, it would sink and ponds, lakes, rivers and even the ocean would freeze solid thereby making life impossible in teh water. Since scientists believe that life first originated in the oceans, this would mean that early life could not have developed and moved to onto land. Additionally, it is the life in the water that sustains life on land, through the oxygen producing algae to the food sources such as fish.
When a body of water freezes on the surface, the ice acts as an insulator to prevent the water below it from freezing. This protects aquatic organisms so that they can survive the winter.