Acids and Bases
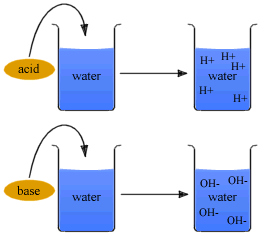
When water ionizes, it loses an equal number of hydrogen ions (H+) and hydroxide ions (OH-). Only a few water molecules at a time dissociate, so the actual numbers of H+ and OH- in water are very small.
Acidic solutions are substances that dissociate in water, releasing H+ ions. The acidity of a substance depends on how fully it dissociates in water. Examples of acidic solutions are lemon juice, vinegar, tomatoes and coffee.
Basic, or alkaline, solutions are substances that either take up H+ ions or release OH-. Milk of Magnesia and ammonia are common basic solutions.
Acidic solutions are substances that dissociate in water, releasing H+ ions. The acidity of a substance depends on how fully it dissociates in water. Examples of acidic solutions are lemon juice, vinegar, tomatoes and coffee.
Basic, or alkaline, solutions are substances that either take up H+ ions or release OH-. Milk of Magnesia and ammonia are common basic solutions.